DF Literature Monitor is an AI-powered solution automating comprehensive literature monitoring for safety vigilance in pharmaco/cosmetovigilance, ensuring compliance and efficiency.
Vendor
datafoundry.ai
Company Website
The field of pharmaco/cosmetovigilance faces significant challenges in monitoring the ever-increasing volume of literature. These include the need for efficient and accurate identification of relevant information, compliance with stringent regulatory requirements, and the substantial strain on resources due to time-consuming, resource-intensive manual screening processes. Additionally, ensuring real-time alerts and fostering effective collaboration among team members can be difficult. DF Literature Monitor transforms literature monitoring for safety vigilance by leveraging state-of-the-art AI models to automate the review of vast amounts of literature. The solution employs semantic search to retrieve relevant articles simultaneously from all major global and local literature databases. It features automated de-duplication, precise extraction of minimum safety information, and auto-population of safety case forms. Users can submit case forms to Safety Databases in E2B or Excel/PDF formats with a single click. The system also provides automated translation of abstracts and full articles to English, enabling pharmacovigilance teams to save up to 60% or more time and effort while ensuring quality and compliance. Recognizing the critical importance of collaboration in pharmacovigilance, DF Literature Monitor includes a configurable workflow to facilitate seamless team collaboration and robust quality control. It is designed as a modular and scalable solution, capable of integrating with leading safety databases and signal management products. This advanced AI-powered solution optimizes safety vigilance processes, leading to better and faster patient safety outcomes by significantly improving the efficiency and accuracy of identifying crucial safety information.
Features & Benefits
- Integrated Literature Sources
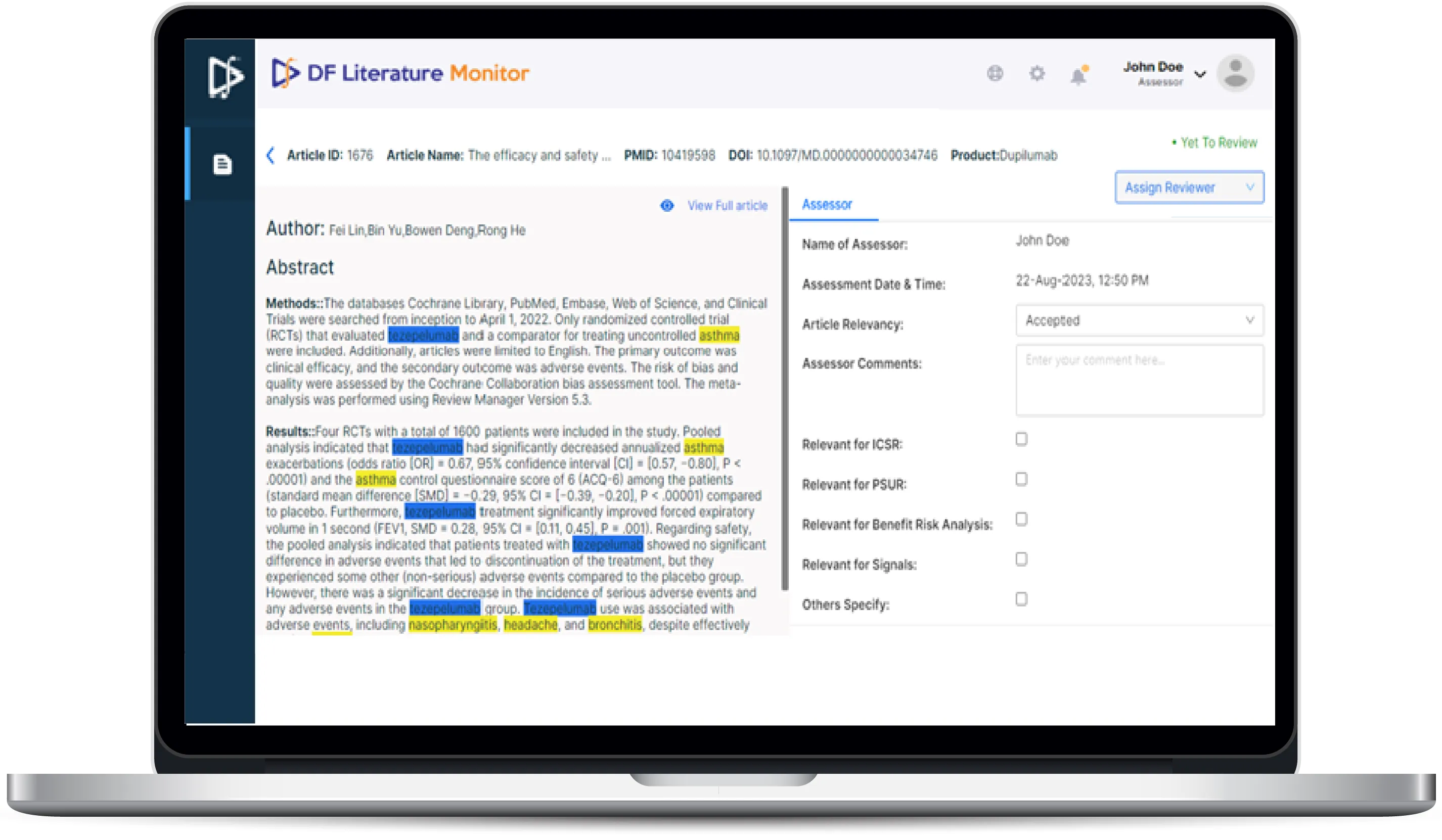
- Integrated with all major global and local literature sources for simultaneous article download.
- Relevancy Ranking
- Articles are ranked based on relevancy, considering potential adverse events (AE) and date of publication.
- AI-Powered Information Extraction
- Utilizes NER (NLP) models for highlighting safety-related information, extracting safety entities, and auto-populating safety case forms, including entity tagging.
- Automated Data Management
- Performs auto de-duplication and automated abstract/article translation into English.
- Configurable Workflows
- Offers configurable workflows, alerts, and notifications to facilitate collaboration and quality control.
- Enhanced Efficiency & Accuracy
- Improves literature monitoring process efficiency by up to 5X and accuracy, leading to up to 60% savings in time and manual effort. NER-based highlights ensure accurate extraction of minimum safety information, reducing errors.
- Regulatory Compliance
- Ensures compliance by extracting data into suitable AE forms for upload to safety databases in E2B R2/R3 format.
- Customization
- Allows customization via an admin module for workflow and product configuration to meet unique pharmacovigilance needs.
- Cost-Effectiveness
- Reduces costs significantly for pharmacovigilance departments through automated processes and enhanced efficiency.
- Multilingual Support
- Removes language barriers with automated translation of articles into English, enabling monitoring of literature in multiple languages.
- Zero Duplication
- Ensures information is not duplicated through automated duplicate search, saving time and effort.