
Annalise Enterprise CXRAnnalise.ai
Developed by clinicians for clinicians, our AI decision support solution can detect up to 124 findings on chest X-rays in seconds. Improves accuracy by 45%, averaged across all analysed findings.
Vendor
Annalise.ai
Company Website
Product details
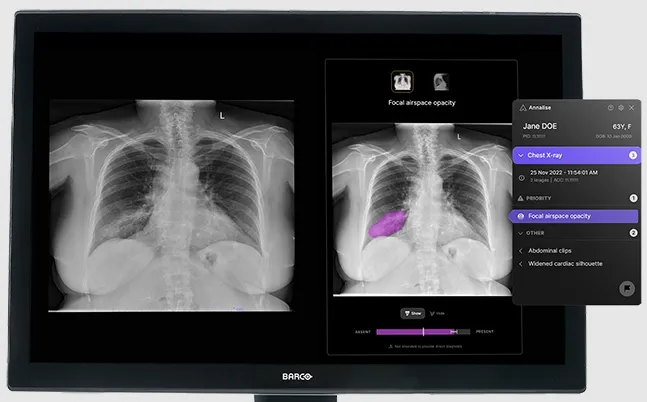
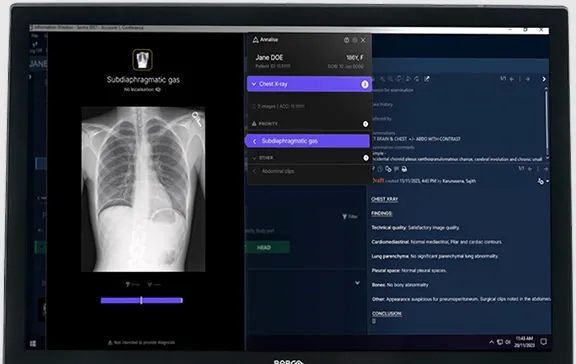
Annalise Enterprise CXR is an advanced AI decision-support solution developed by clinicians for clinicians. It analyzes chest X-rays and detects up to 124 findings in under 20 seconds, acting as a second pair of eyes to enhance diagnostic accuracy and confidence. The solution is CE marked as a class IIb medical device and is available for clinical use in multiple regions including the EU, UK, Australia, India, and more.
Features
- Detection of 124 Findings: Covers a wide range of clinically relevant chest X-ray abnormalities.
- Rapid Analysis: Results available in less than 20 seconds.
- Multiview Correlation: Supports analysis of up to three images per study.
- Confidence Bar: Displays likelihood and uncertainty of findings to aid interpretation.
- Automated Draft Reporting: Generates editable reports integrated into routine workflows.
- Seamless Integration: Customizable UI integrates with PACS and RIS systems.
- Intuitive Design: Most clinicians feel comfortable using it after fewer than 10 studies.
- Extensive Training Dataset: Trained on 782,000+ unique CXR studies from diverse sources.
- Expert Annotation: Annotated by over 150 qualified radiologists using standardized procedures.
- Robust Labeling: Over 280 million CXR labels used for model training.
Benefits
- Improved Accuracy: Average 45% improvement across all analyzed findings.
- Greater Efficiency: 12% average improvement in reporting efficiency.
- Enhanced Confidence: Reduces fatigue and cognitive bias during interpretation.
- Better Patient Outcomes: Early detection of critical conditions like lung cancer.
- Clinician-Endorsed: Widely praised for usability and impact on care quality.
- Global Availability: CE marked and available in Australia, New Zealand, EU, UK, India, ASEAN*, UAE, and more.
- Scalable Deployment: Easily integrates into existing radiology workflows.
Find more products by industry
Health & Social WorkProfessional ServicesInformation & CommunicationView all