Aligned ElementsAligned
Aligned Elements enhances medical device compliance through streamlined design controls and documentation.
Vendor
Aligned
Company Website
Product details
Aligned Elements is a software solution designed specifically for managing the lifecycle of medical devices. It focuses on reducing documentation time and ensuring compliance with regulations such as CE Mark, FDA, and ISO standards. The software integrates end-to-end traceability, risk management, and verification processes, making it an essential tool for medical device developers.
Key Features
- Requirements Management: Manage requirements with version control and change impact analysis across the lifecycle.
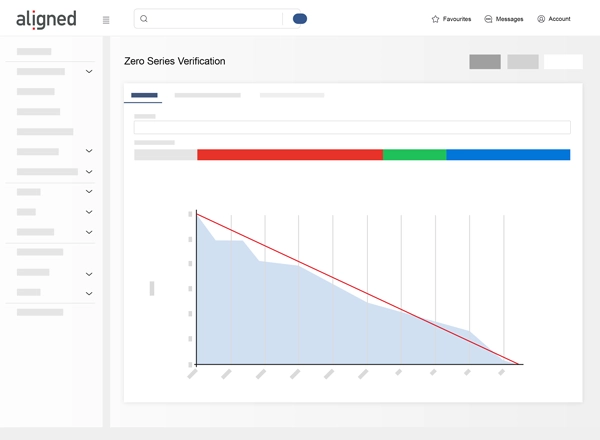
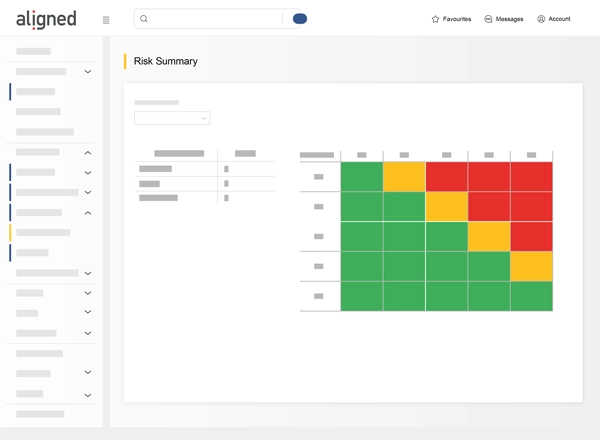
- ISO 14971 Risk Management: Conduct compliant risk assessments with Preliminary Hazard Analysis and FMEA.
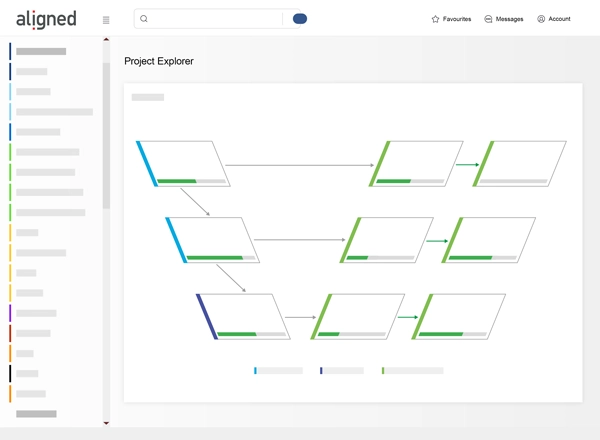
- End-to-End Traceability: Visualize traceability status to eliminate compliance issues.
- Verification & Validation: Run automated and manual test cases with bug tracking integration.
- Document Management: Digitize Technical Files with flexible Word integration.
- Analytics and Visualization: Monitor project gaps and trends.
Benefits
- Improved Efficiency: Reduces time spent on documentation, allowing more focus on innovation.
- Compliance: Ensures compliance with major regulatory standards like FDA QSR and EU MDR.
- Accuracy: Automates consistency monitoring to reduce errors.